Treatment with apraglutide preserves the global homeostatic environment of intestinal microbiota during chemotherapy in mice
Violetta Dimitriadou1.
1Translational Science, VectivBio, Basel, Switzerland
Background: Hematopoietic stem cell transplantation patients experience profoundly altered gut microbiota composition due to dysregulation of intestinal homeostasis by conditioning regimens, broad-spectrum antibiotics, immunosuppressants, and the introduction of foreign lymphocytes from the donor. A growing body of evidence shows reduced microbiome diversity increases the incidence and seriousness of graft versus host disease (GvHD) and bacteremia. Apraglutide, a novel long-acting synthetic glucagon-like peptide 2 (GLP-2), has been shown to protect gastrointestinal (GI) epithelium structure from chemotherapy-induced injury, improve survival, and allowed better body weight maintenance in mice undergoing chemotherapy. The study aimed to evaluate the protective effect of apraglutide on gut microbiota during chemotherapy with cytarabine.
Methods: Balb/c mice received 30 mg/kg of cytarabine on Days 5-9 and apraglutide 3.3 mg/kg on Days 1-18. Control mice received the vehicle on Days 1-18. Fecal samples were collected over 24 hours for bacterial phenotyping at pre-treatment and the day before scheduled termination and for found dead or pre-terminally euthanized animals. Microbiota composition was determined by 16S taxonomical meta-sequencing.
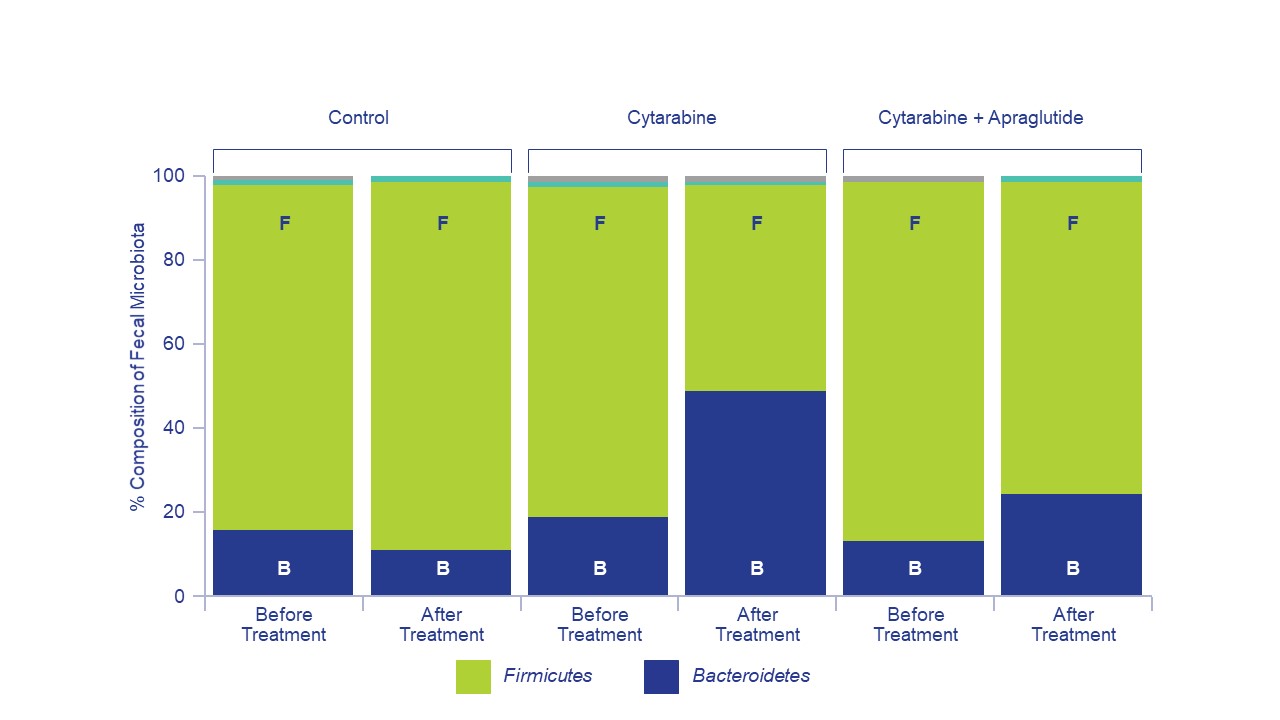
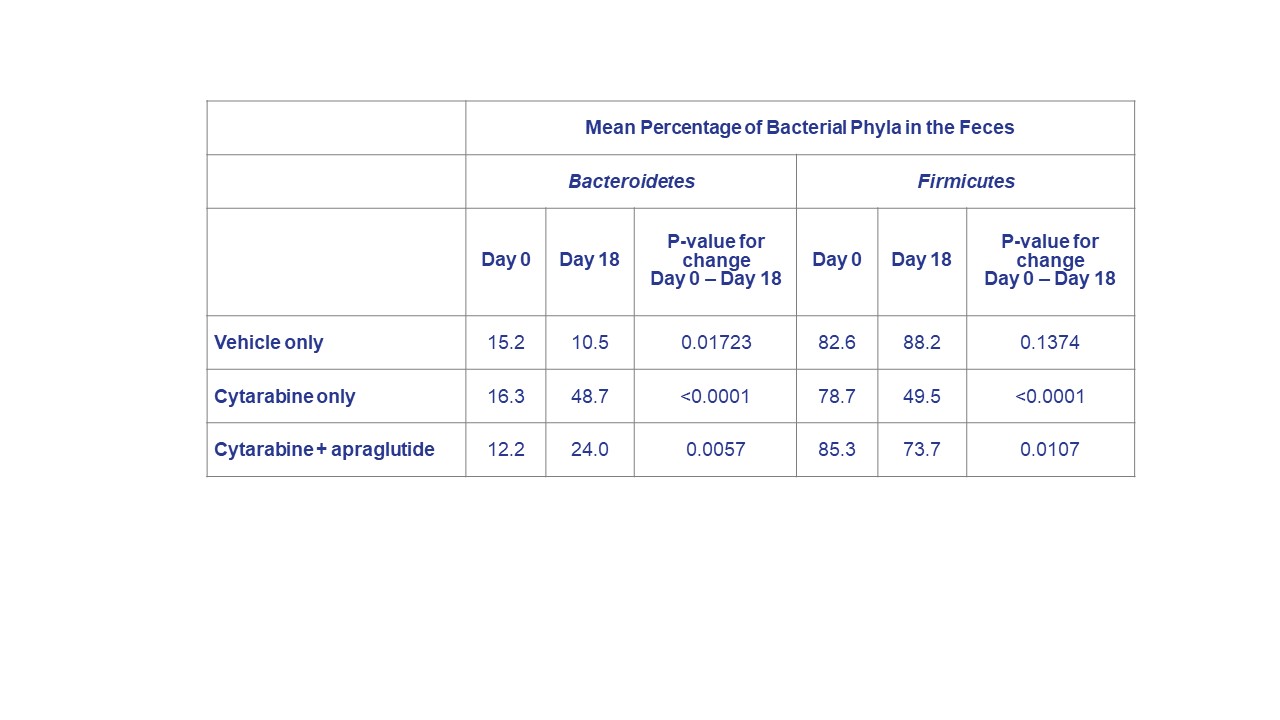
Results: Bacteroidetes and Firmicutes were the two leading bacterial phyla identified. Chemotherapy with cytarabine caused significant changes in the composition of bacterial species, increasing the Bacteroidetes population and decreasing the proportion of Firmicutes bacteria. The change in Bacteroidetes and Firmicutes bacteria levels from Days 0 to 18 was significantly greater in the cytarabine-only and cytarabine + apraglutide mice vs. vehicle. However, this effect was reduced by apraglutide co-administration. The difference in the change between cytarabine-only and cytarabine + apraglutide groups reached statistical significance for both Bacteroidetes (0.2486; p<0.0001) and Firmicutes (0.2037; p<0.0001). In addition, the ratio of Bacteroidetes to Firmicutes bacteria present remained more constant in cytarabine + apraglutide than in the cytarabine-only group.
Conclusions: Chemotherapy profoundly impacted bacterial homeostasis in the mouse intestine, with a notable increase in opportunistic pathogenic bacteria populations. The proportions of different bacterial phyla in feces remained closer to normal when apraglutide was co-administered with chemotherapy. Treatment with apraglutide resulted in the preservation of the global homeostatic environment of the intestinal microbiota. Prevention of intestinal dysbiosis may contribute to the improved outcomes (reduced body weight loss, increased survival) observed in mice when apraglutide is administered concomitantly with chemotherapy agents.

right-click to download
