Analysis of immune cells draining from the abdominal cavity as a novel tool to early prediction of clinical events. Prospective multicenter study - INIGMA follow-up report
Maria Virginia Gentilini1,2, Hector Solar1, Carolina Rumbo1, Javier Serradilla 3, Francisco Hernández Oliveros 3, Gustaf Herlenius 4, Mihai Oltean4, Laurence Ceulemans 5, Jacques Pirenne5, Martín Rumbo6, Gabriel Eduardo Gondolesi1,2.
1Multiorgan Transplant Institute, Nutritional Support Unit, Intestinal Rehabilitation and Transplanta, Favaloro University Hospital, Caba, Argentina; 2Translational Research Laboratory and Immunology associated to Transplantation, IMETTyB- CONICET, Favaloro University , Caba, Argentina; 3Pediatric Surgery Service, La Paz University Hospital, Madrid, Spain; 4Department of Surgery, Institute for Clinical Sciences, University of Gothenburg, Gothenburg, Sweden; 5Department of Abdominal Transplantation Surgery, University Hospital Leuven, Leuven, Belgium; 6Immunology and Pathophysiology Studies Institute, School of Exact Sciences, National University of La Plata, La Plata, Argentina
Introduction: International Network for Intestinal Graft Monitoring and Analysis – INIGMA” project is a multicenter study started in 2015. The rationale of this work consists in that the abdominal drainage fluid collected after intestinal transplant (ITx) contains immune cells trafficking from the implanted intestine, and changes of immune cell composition, especially increase of neutrophils, correlated with the appearance of future clinical events (rejection, infection, or other events). The main objective of this project is to validate this non-invasive method for predict the development of abdominal complications after the first post-op days, based on the analysis of draining cell composition and their correlation with clinical follow-up. Here, we aimed to present a follow-up of our study.
Material and Methods: This prospective observational study started in 2008 by Favaloro Foundation University Hospital, Argentina and turned into a multicenter study involving the University of Gothenburg, Sweden; University of Leuven, Belgium and La Paz University Hospital, Spain from 2015 to 2022. The cell composition of the abdominal draining fluid and blood collected during the first post-op days was analyzed by differential cell counter and was correlated with the clinical outcome.
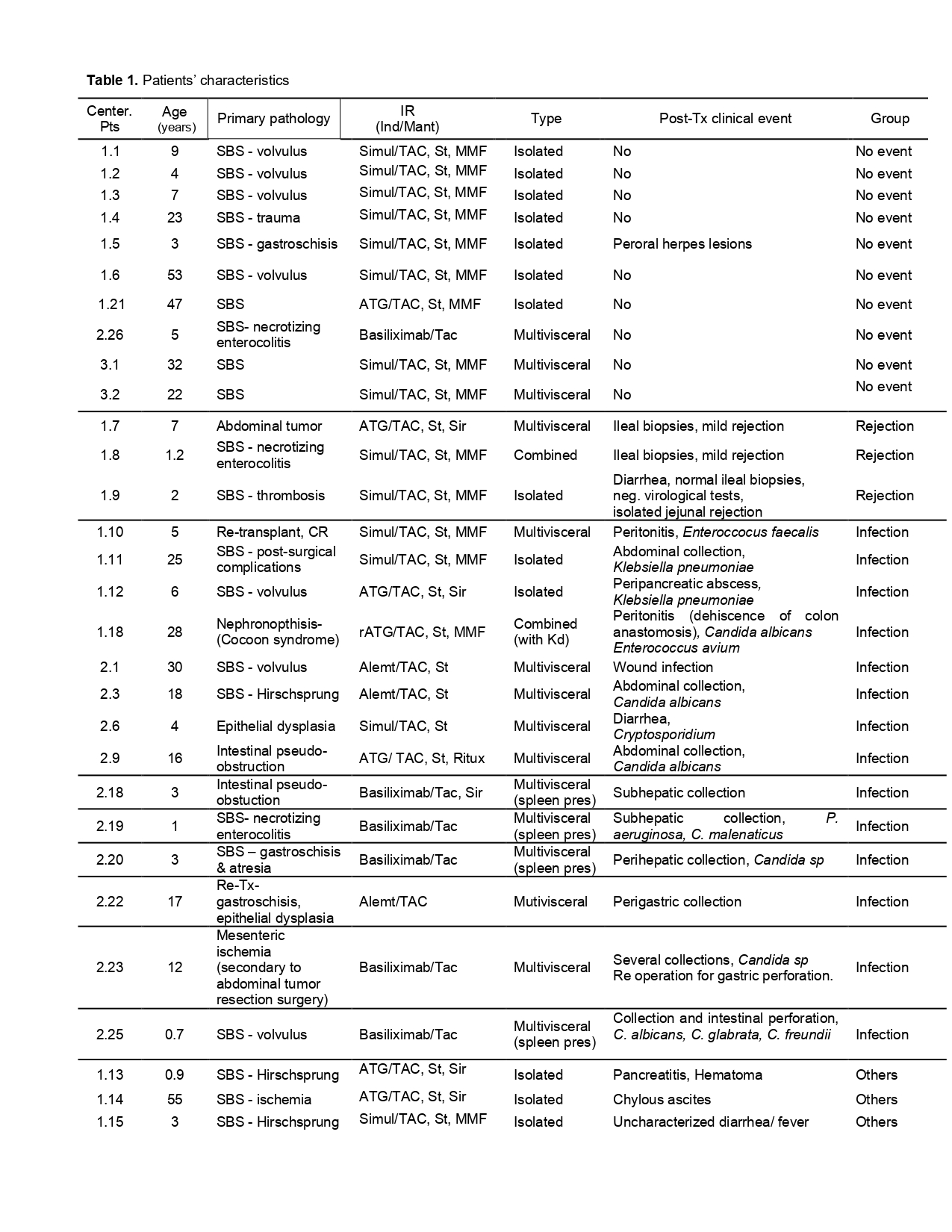
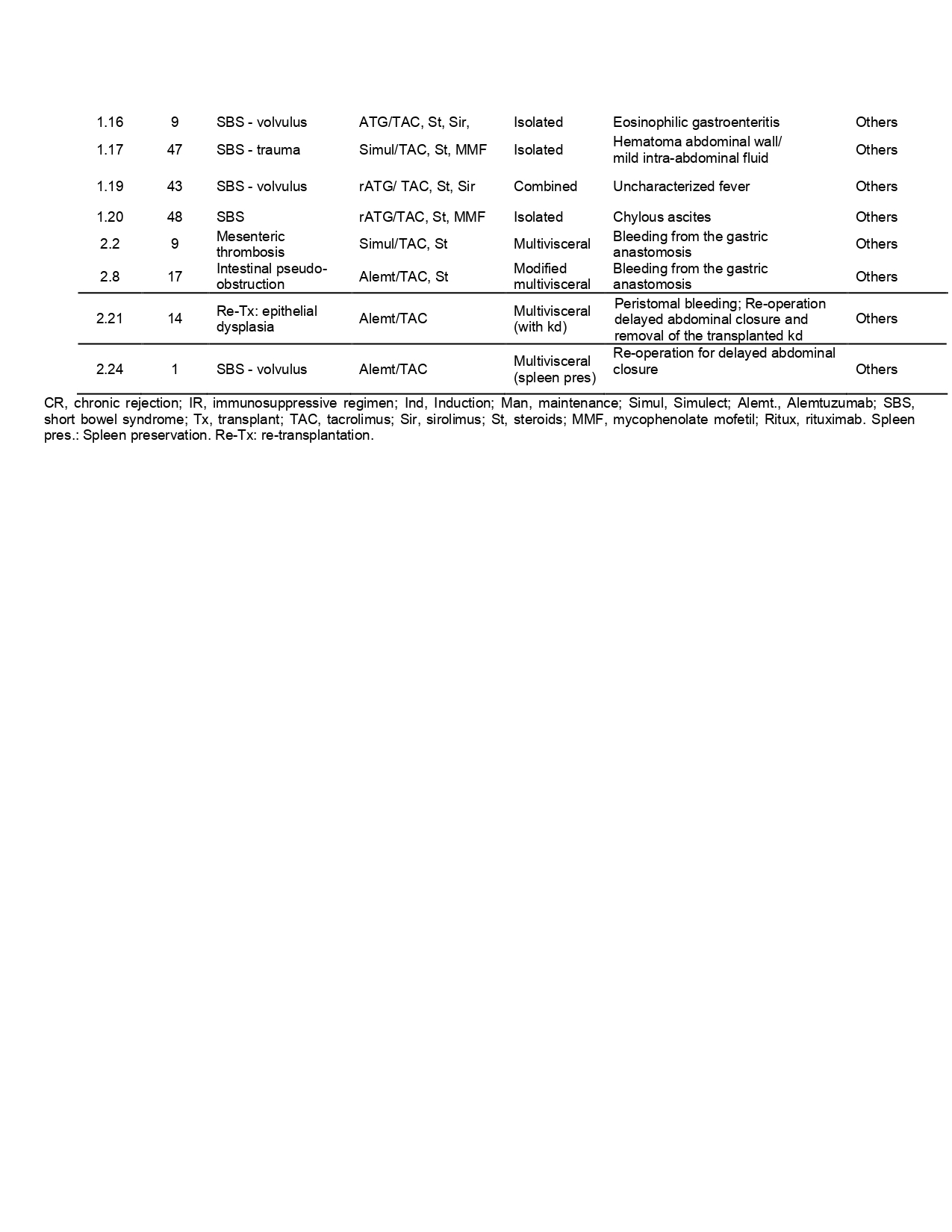
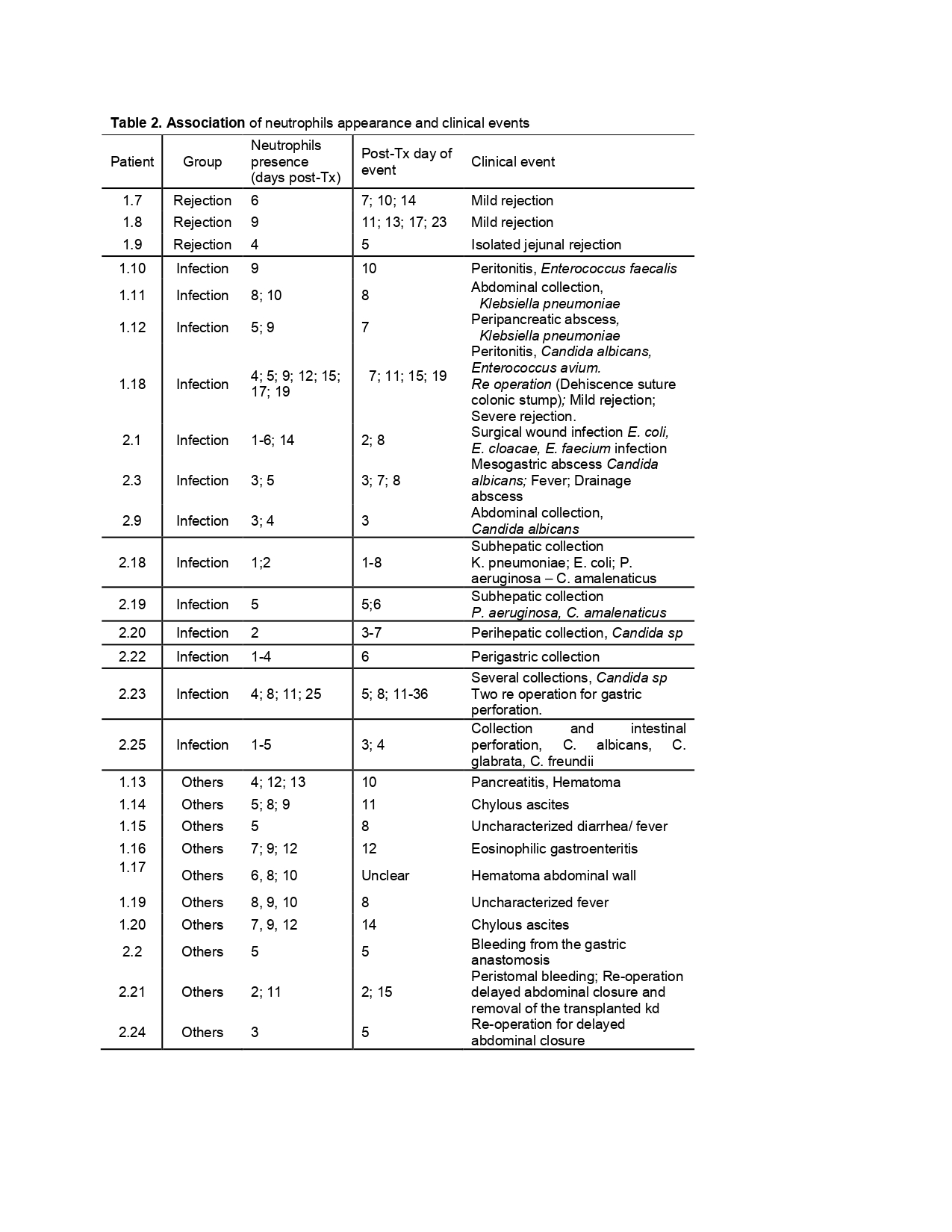
Results: A total of 38 patients (pts) with complete biochemical and clinical information were enrolled by 3 of the 4 centers (Table 1). Blood samples showed a predominance of neutrophils and marked leukopenia, independently of the age, immunosuppressive protocol used, and clinical event reported. Whereas draining cell composition showed neutrophilic predominance when patients suffered intra-abdominal clinical complications that change to a leukocyte prevalence when the clinical event resolved. When a new shift to a neutrophil dominant content is observed in the drainage, it anticipates the development of a clinical event in the peritoneal cavity (26/38 pts). Table 2 summarizes the association between neutrophils and clinical events. Sensitivity=92,9%; Specificity=80%; PPV=92.9%, NPV=80%.
Conclusion: our preliminary results suggest that abdominal clinical events can be early predicted after ITx by analyzing the changes in the drainage cell composition, particularly when a shift to a neutrophilic dominance is registered. Thus, cell counts from the drainage should be included as part of the daily evaluation of pts receiving an ITx. This analysis provides further support to use this non-invasive approach to monitor the grafts and the transplanted pts providing easily information for clinical management decisions. A larger pts cohort should be required to validate this methodology.

right-click to download
