Delta changes in donor-derived cell-free DNA (dd-cfDNA) complement the donor fraction in kidney transplant surveillance
Alexander Wiseman1, Gaurav Gupta2, Muralidharan Jagadeesan3, Vinaya Rao7, Mohanram Narayanan5, Nikhil Agrawal6, Grigory Shekhtman6, Yi Fu6, Kevin Pinney6, Sanjiv Anand4.
1Medicine, Centura Health-Porter Adventist Hospital, Aurora, CO, United States; 2Medicine, Virginia Commonwealth University, Richmond, VA, United States; 3Medicine, George Washington School of Medicine, Washington DC, DC, United States; 4Medicine, Intermountain Healthcare, Murray, UT, United States; 5Medicine, Baylor Scott and White, Austin, TX, United States; 6Kidney Transplant, CareDx, Brisbane, CA, United States; 7Medicine, Medical University of South Carolina, Charleston, SC, United States
KOAR registry.
Introduction: Delta changes in dd-cfDNA levels over time may identify patients with evolving alloimmune injury, providing complementary information to thedonor fraction. We explored the dd-cfDNA trajectories preceding biopsy-proven rejection (BPAR) events among kidney transplant recipients enrolled in theKidney allograft Outcomes AlloSure Registry (KOAR, NCT03326076) and undergoing longitudinal surveillance with dd-cfDNA.
Methods: In the primary analysis, we compared the change in dd-cfDNA from baseline to the time of BPAR (for-cause or surveillance biopsies). We identifiedpatients with first-time rejection events and at least 3 prior dd-cfDNA results; the index result was obtained ≤30 days before BPAR; we then selected the lowestof two preceding results as the patient-specific baseline and compared this with the index result. We then analyzed patients with rejection (first or subsequent, atleast 90 days apart) and ≥2 dd-cfDNA results, including one within 30 days of the index biopsy. The reference change value (RCV) was calculated using areference population of patients with stable allograft function, at least 3 dd-cfDNA measurements, and no significant clinical events.
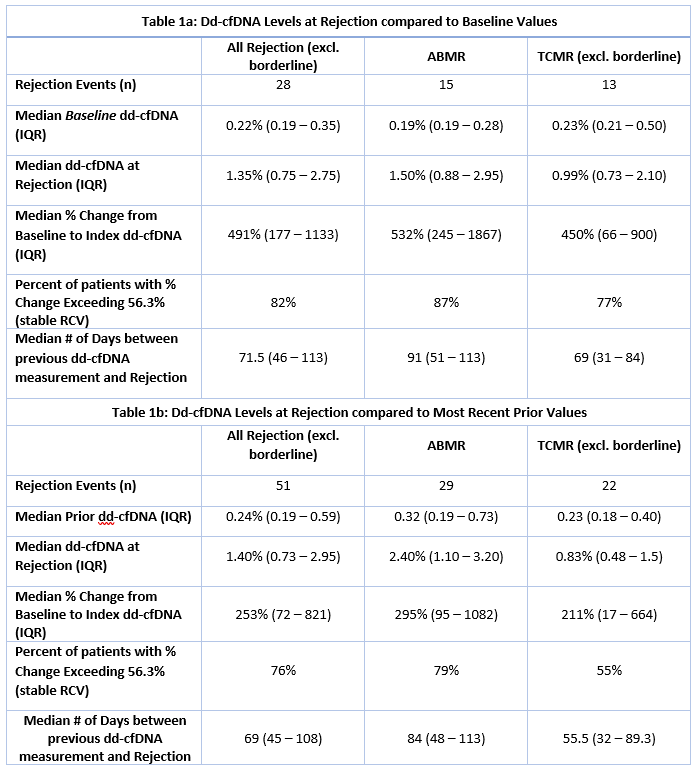
Results: A total of 28 patients with BPAR met criteria for the primary analysis; among these, median baseline dd-cfDNA was 0.22% (IQR: 0.19 - 0.35) and mediandd-cfDNA at the time of rejection was 1.35% (IQR: 0.75 - 2.75), representing a 491% (IQR: 177 - 1133) increase between these results, obtained 71.5 (IQR: 46 -113) days apart (Table 1A).
51 events met criteria for the second analysis where a median increase of 253% (IQR: 72 - 821%) between sequential dd-cfDNAvalues obtained 69 days (IQR: 45 - 108) apart preceded biopsy-proven rejection events (Table 1B). The calculated RCV for the stable reference population withinKOAR was 56.3%. 39 of these 51 events (76%) demonstrated increases greater than this RCV.
Conclusions: Longitudinal surveillance with dd-cfDNA allows the integration of delta changes and trajectories over time, allowing earlier identification ofevolving allograft injury. Significant changes from an established baseline and between sequential values ahead of biopsy-proven rejection highlight how longitudinal surveillance may improve diagnostic performance.

right-click to download
