
Modern medicine needs modern technology tools
Zohreh Soltani1, Sepehr Soltani 2, Emily Johnson1.
1Nephrology, Ochsner Health, New Orleans, LA, United States; 2Alborz University of Medical Science, Karaj, Iran (Islamic Republic of)
Introduction: Early detection of subclinical rejection is critical to prevent the allograft loss. Currently used methods to detect subclinical rejection include surveillance biopsy protocol, serum creatinine level, eGFR or DSA; however, these methods have limited sensitivity and specificity to accurately detect subclinical rejection. Donor derived cell-free DNA (dd cf-DNA) is a non-invasive biomarker for allograft injury and has been shown to predict allograft rejection with higher sensitivity to detect subclinical rejection compare to some methods (1,2). Here, we show the value of dd cf-DNA in detecting early allograft rejection by presenting two cases where dd cf-DNA assay, AlloSure (CareDx) detected subclinical rejection while other indicators showed stable allograft function.
Methods: Ochsner uses AlloSure (CareDx) for a surveillance for the high-risk patients.
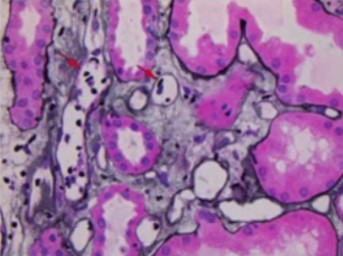
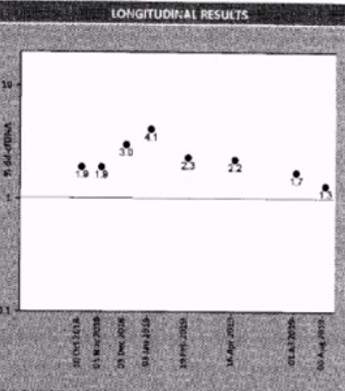
Results: Thirty-two years old Caucasian female with end stage kidney disease secondary to HIV infection underwent deceased-donor renal transplant in 2018 with KDPI 19% and cPRA 84%. Serum creatinine level had been stable at 1.1mg/dL to 1.2mg/dL post-transplant. This patient was monitored using AlloSure (CareDx) for 1-,2-,3-,4-,6- and 9-month post-transplant. AlloSure values for month 1 to 6 were <0.21, which indicates stable allograft function. However, at 9-month post-transplant, AlloSure value went up to 4.5%, which indicates possible allograft injury, while serum creatinine level remained stable at 1.2 mg/dL with low level of class 2 DSA (DR-1573). Despite stable creatinine level and low level DSA, we initiated biopsy because of sharp increase in AlloSure value. Biopsy revealed acute T-cell mediated rejection and acute vascular rejection. The second case is a 33 years-old Caucasian female with history of previous renal transplant. The first allograft failed in 2018 due to renal artery and vein thrombosis. The patient underwent deceased donor kidney transplant in 2019. Serum creatinine level was stable at 1.0 – 1.3 mg/dL and sharp increase in respectively. At month 3, AlloSure value went up to 0.78% but DSA was negative. We initiated biopsy based on increase in AlloSure value and it revealed acute antibody mediated rejection (ABMR) (Figure 1). The third case is a 56 years-old male with history of end-stage renal disease secondary to hypertension. The patient received deceased donor kidney transplant in 2017. He developed acute renal failure (Cr went up from 1.2 mg/dl to 1.7 mg/dL) few months after kidney transplant and his kidney biopsy showed acute antibody mediated rejection and T-cell mediated rejection which were treated. At 1-year post-transplant, serum creatinine level remained 1.2 mg/dL to 1.3 mg/dL. He was place on monthly Allosure surveillance. The first value was 1.9%, which indicate possible allograft injury. Monthly monitoring showed increase trend in AlloSure value up to 4.1% while Cr remained stable at 1.2 mg/dL-1.3 mg/dL. At that point, biopsy was performed, and it revealed acute antibody mediated rejection and chronic active antibody rejection. Patient was treated with for ABMR. In addition, monthly Tocilizumab was considered for chronic active antibody mediated rejection for 6 months. AlloSure values reflected the early detection of rejection in a patient with stable Cr as well as the efficacy of treatment with down trending values (Figure 2).
Conclusion: For all 3 cases, biopsy would not have been initiated most likely due to stable creatinine level and weak or negative DSA. If this had gone untreated for long period of time, this would have led to significant decrease in graft survival. These cases demonstrate that normal serum creatinine does not mean patient has no active inflammatory process. When other indicators are normal/stable, AlloSure was able to detect early rejection. All three cases demonstrated the value of AlloSure as a monitoring tool for detection of early allograft rejection.
References:
Snyder TM, Khush KK, Valantine HA, Quake SR: Universal noninvasive detection of solid organ transplant rejection. Proc Natl Acad Sci USA 108: 6229–6234, 2011
Cell-Free DNA and Active Rejection in Kidney Allografts Roy D. Bloom, Jonathan S. Bromberg et al, J Am Soc Nephrol 28: 2221–2232, 2017

right-click to download
