Real-world treatment patterns and outcomes for cytomegalovirus infection and disease in solid organ transplant recipients in selected countries outside North America and Europe: a systematic review
Jinzhen Cai1, Monica Slavin2, Inderjeet Singh3, Anudeep Sandhu4, Dirk Demuth4, Helio Tedesco-Silva5.
1The Affiliated Hospital of Qingdao University, Qingdao, People's Republic of China; 2Victorian Infectious Diseases Service, Royal Melbourne Hospital, Melbourne, Australia; 3Takeda Pharmaceuticals India Pvt Ltd, Gurgaon, India; 4Takeda International AG – Singapore Branch, Singapore, Singapore; 5Division of Nephrology, Hospital do Rim, Universidade Federal de São Paulo, São Paulo, Brazil
Background: In Europe and North America, ganciclovir (GCV) and valganciclovir (VGCV) constitute the standard of care for the management of cytomegalovirus (CMV) infection and disease in solid organ transplant (SOT) recipients. As practices vary across countries due to differences in access to health care or resources, a systematic review was undertaken to evaluate treatment patterns for CMV infection and disease post-SOT in selected countries outside of Europe and North America and describe related outcomes.
Methods: Information sources (search period: 01 Jan. 2011 – 21 Jul. 2021) included literature search (Ovid® MEDLINE and Embase, Cochrane Database of Systematic Reviews and World Health Organization database Global Index Medicus), pragmatic searches of the grey literature, and snowballing of references lists. Observational studies that included SOT (any organ) recipients (any age) who developed CMV infection or disease in 15 selected countries in Asia-Pacific, Latin America, Russia and the Middle East and reported on the outcomes of interest (treatment patterns for CMV infection and CMV disease, proportion of patients (pts) with resistant and/or refractory CMV including definitions, treatment-related outcomes and adverse events, AEs) were included. The protocol was registered in PROSPERO (CRD42020205559).
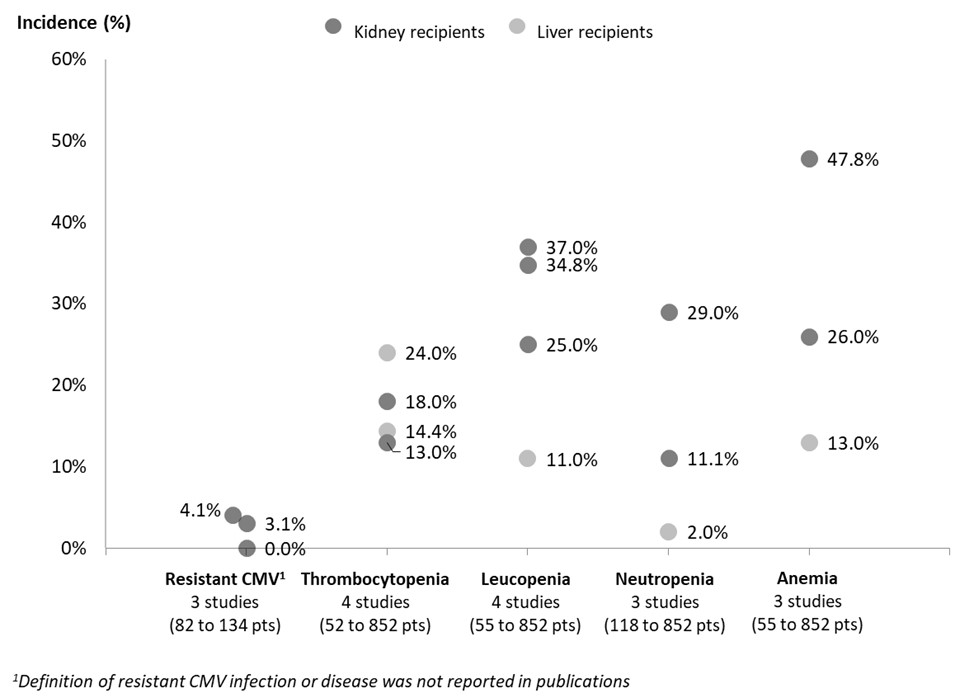
Results: A total of 23 studies (18 to 1,620 pts), most (87.0%, n=20) conducted in adults (≥ 18 years), were included (kidney: n=19 [82.6%]; liver: n=3 [13.0%]; mixed SOTs: n=1 [4.3%]). In studies reporting on preventive strategies used for CMV (n=4), similar proportions of pts receiving prophylaxis and pre-emptive therapy were found (45% to 58.7% pts and 41.3% to 56.3% pts, respectively). Intravenous (IV) GCV and VGCV were the preferred first-line (1L) treatments for both prevention (IV GCV: 77.8%-100% pts, VGCV: 44.4%-99.2% pts) and treatment (IV GCV: 77.8%-100% pts) of CMV infection and disease. In 3 studies, the treatment duration for CMV disease was between 12 and 90 days. In pediatric pts who received GCV as pre-emptive therapy after prophylaxis, time to viremia clearance was 3.5 months. Rates of CMV resistance were reported in 3 studies (Figure 1). No data on risk factors for CMV resistance nor on pts with refractory CMV were found. Hematological AEs (i.e., thrombocytopenia, leucopenia, neutropenia, anemia) were reported in up to 25% of pts receiving IV GCV, as prophylaxis or pre-emptive therapy (Figure 1). Data stratified according to treatment strategy were not available.
Conclusion: The conventional 1L treatment for both prevention and treatment of CMV was IV GCV or VGCV. However, as identified studies reported that 1 in 4 pts experience hematological AEs with these therapies, there remains an unmet need for this patient population. Data on the proportion and management of SOT pts with resistant and/or refractory CMV remains scarce in countries outside of Europe and North America.
This review was funded by Takeda International AG – Singapore Branch. JC has received grants from Takeda company. MS has received grants from F2G, Gilead, Merck and personal fees from Gilead, Pfizer, Takeda and Roche for work outside of this research. IS is an employee of Takeda Pharmaceuticals India Pvt Ltd. AS and DD are employees of Takeda International AG – Singapore Branch and hold stock options. HT has received research grants to conduct clinical trials from Novartis, Pfizer, BMS, Natera, and Astra Zeneca. Literature retrieval, analysis and medical writing support were provided by Aurore Bergamasco, Camille Goyer, and Yola Moride of Yolarx Consultants and funded by Takeda International AG – Singapore Branch.

right-click to download
